Leucovorin (Folinic Acid) and Autism: What the FDA Actually Did—Mechanism, Evidence, and Who Might Benefit
Key takeaway: Viral posts claiming the FDA “approved the first medication for autism” are misleading. The FDA action in 2025 centered on making leucovorin calcium available/authorized for cerebral folate deficiency (CFD)—a rare neurological condition that can present with autistic features—not a universal treatment for autism spectrum disorder (ASD). FDA press release
What is leucovorin?
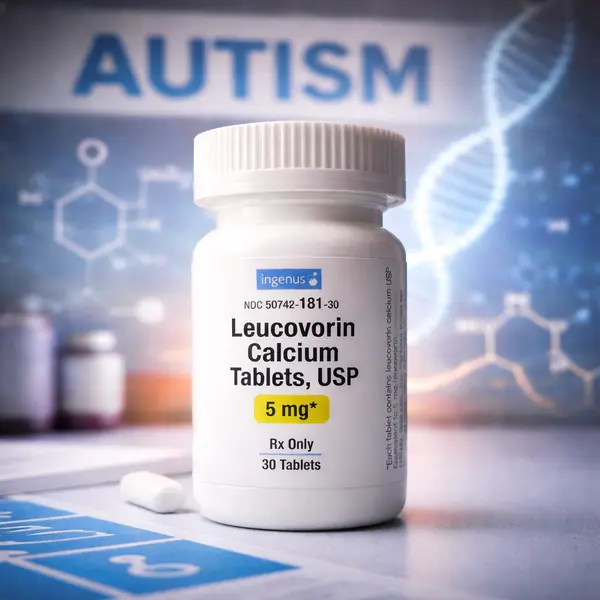
Leucovorin (also called folinic acid) is a prescription form of reduced folate related to vitamin B9. It has long been used in medicine—most commonly as “leucovorin rescue” to reduce toxicity from folate-antagonist drugs such as methotrexate, and in some chemotherapy regimens. Its standard, long-standing labeled uses are not “autism.” DailyMed (FDA label listing)
What did the FDA do in 2025—and what did it NOT do?
In September 2025, the FDA announced actions to make leucovorin calcium tablets available for patients with cerebral folate deficiency (CFD), a disorder involving impaired folate transport into the brain. CFD can include developmental delays and autistic-like symptoms, which helped drive headlines and social media claims. FDA announcement
However, major pediatric guidance emphasized a crucial distinction: this is not an FDA approval for “autism in general.” The American Academy of Pediatrics (AAP) notes that evidence is still limited, that a large phase 3 multicenter trial has not been completed, and that any prescribing should involve careful shared decision-making and monitoring. AAP FAQ
How leucovorin could work in a subset of autistic children
The biological rationale focuses on brain folate availability and one-carbon metabolism (a network of reactions involved in methylation, neurotransmitter synthesis, and DNA/RNA processes). Unlike folic acid, folinic acid is already in a reduced form and can be converted into active folate cofactors more directly.
1) Cerebral folate deficiency (CFD) and folate transport problems
In CFD, folate levels in the central nervous system are low despite normal or near-normal blood folate, typically due to impaired transport across the blood–brain barrier. One proposed mechanism in some patients involves folate receptor alpha (FRα) autoantibodies, which may interfere with folate entry into the brain. The FDA’s 2025 focus on CFD is grounded in this transport biology. FDA announcement
2) Why this matters for symptoms
If low brain folate contributes to language, attention, or social communication difficulties in a subgroup, restoring folate-dependent pathways could plausibly lead to improvement. Importantly, this is a subset hypothesis—not a one-size-fits-all autism treatment.
What improvements have studies reported?
Clinical studies of folinic acid/leucovorin in ASD have most consistently explored outcomes related to language and communication, with some studies also examining broader autism symptom scales. Results vary, and study sizes are generally modest.
- Verbal communication gains (language-focused outcomes): A randomized, double-blind, placebo-controlled trial reported improvement in verbal communication in children with autism and language impairment receiving folinic acid. Molecular Psychiatry (2018 trial)
- Symptom scale improvements in some RCT data: A 2024 double-blind RCT evaluated folinic acid (up to 50 mg/day) over 24 weeks and reported changes on autism rating scales compared with placebo in children receiving standard care. PubMed (2024 RCT)
- Additional randomized trial evidence (shorter duration): A 2025 randomized clinical trial evaluated high-dose folinic acid in children with ASD and reported on safety/efficacy outcomes. Nutrients (2025 trial)
Reality check: Even supportive findings are often described as modest and most relevant to select groups (for example, children with markers consistent with folate transport or metabolism issues). The AAP highlights the lack of large phase 3 trials and the need for more rigorous evidence before routine use. AAP FAQ
Important recent development: reporting in early 2026 notes that the largest leucovorin-autism randomized trial was retracted, which further weakens the evidence base and reinforces the need for careful interpretation of “breakthrough” claims. Disability Scoop report (retraction)
Safety, side effects, and why medical supervision matters
Leucovorin is a prescription drug, not just a vitamin supplement. Potential side effects reported clinically can include gastrointestinal upset, sleep disturbance, and behavioral activation/irritability in some children. Pediatric organizations emphasize close monitoring if used off-label for ASD-related symptoms and a careful risk–benefit discussion with a clinician. AAP FAQ
In addition, leucovorin labeling includes important warnings/contraindications in certain anemia contexts (e.g., it is not appropriate therapy for pernicious anemia due to vitamin B12 deficiency, because it may mask hematologic signs while neurologic damage progresses). DailyMed (FDA label listing)
Who is most likely to be considered for leucovorin?
Based on the FDA’s CFD framing and the clinical literature, leucovorin discussions most often arise for children who may have:
- Evidence suggesting cerebral folate deficiency or impaired folate transport
- Language impairment where prior trials have concentrated potential benefits
- Clinical indications for targeted testing (determined by a specialist), rather than broad “autism” alone
The AAP underscores that the science does not support routine use for all autistic children and that more research is needed. AAP FAQ
Final Takeaway
Leucovorin is not a “first cure” or a universal medication for autism. The FDA’s 2025 action was tied to cerebral folate deficiency—a condition that can overlap with autistic symptoms in a subset of patients—and the broader evidence for ASD symptom improvement remains limited and mixed. The most consistent signal across trials has involved language/verbal communication, but recent retraction news and the absence of large phase 3 studies mean families should treat social-media certainty with caution and rely on specialist guidance. FDA announcement AAP FAQ
Editorial Note: Some links on this page may earn us a commission at no extra cost to you.
Our reviews remain independent and evidence-based.